|
9/5/2023 0 Comments Crystalmaker stacking
The Crystal Chemistry library provides a wealth of teaching resources for understanding fundamental "type" structures, such as close packed metals.īuilding Crystals provides a self-guided tour to important structures in terms of their fundamental building bricks and how these are arranged. Quiz: Learn crystallography with these exercises on Lattice Types, Miller Indices and Basic Structure Types. 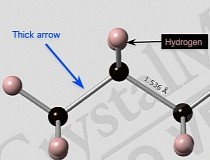
Includes animated teaching models of cubic close packing, hexagonal close packing, diamond and graphite, etc. Includes YBCO (Superconductor) ZIF8 (potential gas-storage phase).Ĭrystal Chemistry: Crystallographic "Type Structures", organised by anion/cation ratios, e.g., "AB, AB2, AB2X4, etc.". New Materials: More complex structures introduced in terms of their fundamental building blocks. Includes: Garnet Pyroxene Amphibole Mica Kaolinite Feldspar Quartz Zeolite ZK5. Silicates: Introduction to Silicate Polymerization Sequence of key silicate minerals illustrating the sequence from isolated tetrahedra (orthosilicates), through to infinite chains, sheets and fully-polymerized frameworks. Includes: Wurtzite Sphalerite Halite Olivine Spinel Hematite. Each multi-structure document provides a unique tour of a key structure, building on concepts such as close-packing, interstices, polymerization and the use of structural motifs to represent coordination units or molecular groups and simplify visualization.Ĭlose Packing: Introduction to Close Packing Hexagonal Close Packing Cubic Close Packing.įilling Interstices: Introduction to Interstices Structures based on filling tetrahedral or octahedral interstices in hexagonal- or cubic-close-packed materials. Polyhedra: Coordination polyhedra found in typical crystals.īuilding Crystals: Self-guided tutorials introducing fundamentals of crystal chemistry. Lattice Defects: 3D models showing twins, polysynthetic twinning, edge & screw dislocations dislocation loop Frenkel, Schottky defects stacking fault. Lattice Types: Rotatable models of each of the 14 Bravais Lattices, plus Hexagonal/Rhombohedral comparison model. Atomic Radii: Periodic Tables of the Elements showing atomic, ionic, covalent and van-der-Waals radii.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
